F_Ingrasciotta
Guest
Good evening to all,
I found myself having to perform an estimate of the time needed to fill a 15-litre cylinder.
Consider filling a volume v cylinder with air, initially under the conditions pi and ti, by means of a compressor with a capacity equal to q_in and t_in temperature. the final pressure is pf. the filling time of the cylinder is formed.
the starting data available are as follows: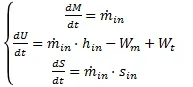
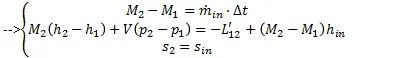 from the last equation I made the following consideration:
from the last equation I made the following consideration: After that I took a table that shows me the value of cp at the temperature range and I linearized between the points known so as to obtain a function of cp(t)
After that I took a table that shows me the value of cp at the temperature range and I linearized between the points known so as to obtain a function of cp(t)
the next step was to calculate the value of t2 in an iterative way starting from the last equation, the process is reported below:
therefore defined the temperature t2 and the pressure p2 it is possible to obtain the m2 mass of air inside the cylinder by means of the perfect gas eq.
the filling time obtained is therefore 3.6 minutes.
I ask your opinion on these considerations, I imagine that in order to have a much more realistic result and that it is not too influenced by the assumptions of departure I have to support a fem to solve the various differentials that are obtained from the budgets.
Should I also discuss the pressure leap to carry out accounts on smaller jumps so as to get more sensible values?
Thank you in advance.
I found myself having to perform an estimate of the time needed to fill a 15-litre cylinder.
Consider filling a volume v cylinder with air, initially under the conditions pi and ti, by means of a compressor with a capacity equal to q_in and t_in temperature. the final pressure is pf. the filling time of the cylinder is formed.
the starting data available are as follows:
- v = 15 l
- pi = 1 bar (initial pressure)
- ti = 20°c (initial time)
- q_in = 170 lt/min (shipport)
- t_in = 20°c (temperatura in mandata)
- pf = 147 bar (final pressure)
- isoentropic process
- adiabatic process
- air considered perfect gas
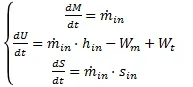
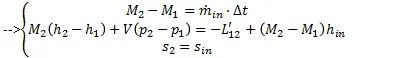 from the last equation I made the following consideration:
from the last equation I made the following consideration:the next step was to calculate the value of t2 in an iterative way starting from the last equation, the process is reported below:
- I place a value of t2 said t2*
- calculation cp(t2*)
- echo t2 from the last equation of entropy using cp(t2*)
- If the percentage error between t2 and t2* is less than 5% I consider the correct calculation, otherwise I select a new value of t2
therefore defined the temperature t2 and the pressure p2 it is possible to obtain the m2 mass of air inside the cylinder by means of the perfect gas eq.
the filling time obtained is therefore 3.6 minutes.
I ask your opinion on these considerations, I imagine that in order to have a much more realistic result and that it is not too influenced by the assumptions of departure I have to support a fem to solve the various differentials that are obtained from the budgets.
Should I also discuss the pressure leap to carry out accounts on smaller jumps so as to get more sensible values?
Thank you in advance.
